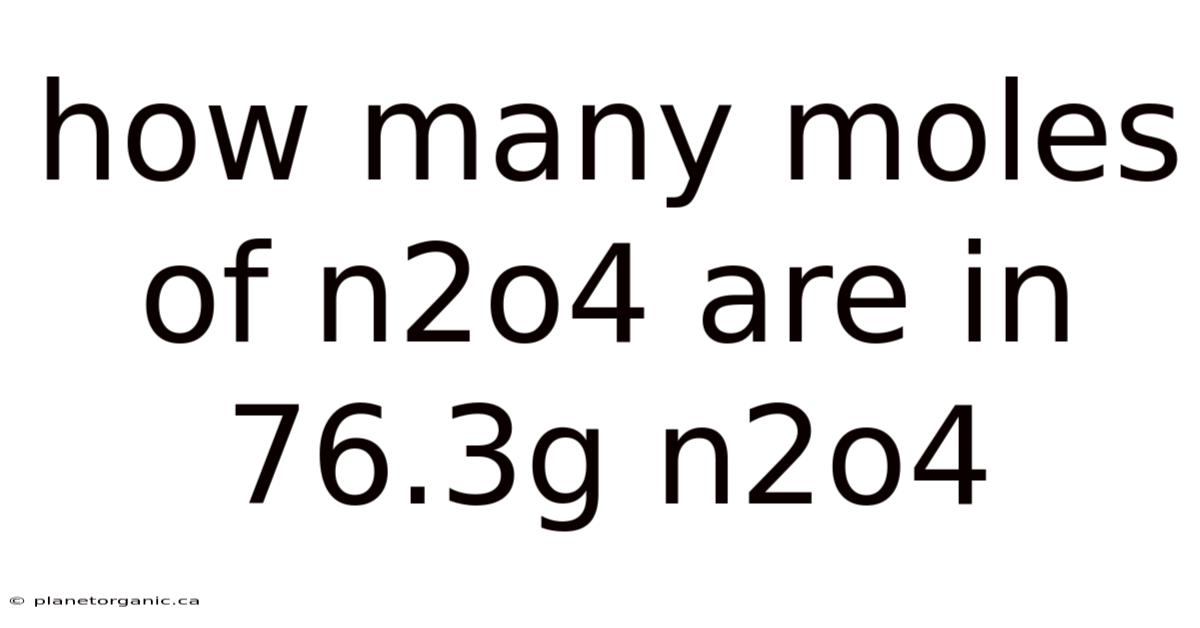The relationship between mass, molar mass, and the number of moles is fundamental to understanding stoichiometry in chemistry. To determine how many moles of dinitrogen tetroxide (N₂O₄) are in a 76.3g sample of N₂O₄, you need to use the molar mass of the compound as a conversion factor.
Understanding Moles and Molar Mass
Before diving into the calculation, let's clarify a few key concepts.
-
Mole: The mole (symbol: mol) is the standard unit of amount in chemistry. One mole contains exactly 6.02214076 × 10²³ elementary entities. This number is known as Avogadro's number (Nᴀ). The mole is essential because it provides a bridge between the atomic scale and the macroscopic scale The details matter here..
-
Molar Mass: The molar mass (M) of a substance is the mass of one mole of that substance, expressed in grams per mole (g/mol). The molar mass is numerically equal to the atomic or molecular weight of the substance in atomic mass units (amu). You can calculate the molar mass of a compound by summing the atomic masses of each element in the compound, as found on the periodic table, and multiplying by the number of atoms of each element in the chemical formula.
Calculating the Molar Mass of N₂O₄
To convert grams of N₂O₄ to moles, the first step is to calculate its molar mass. Dinitrogen tetroxide (N₂O₄) consists of two nitrogen atoms and four oxygen atoms.
-
Identify the Atomic Masses:
- Nitrogen (N) has an atomic mass of approximately 14.01 amu.
- Oxygen (O) has an atomic mass of approximately 16.00 amu.
-
Calculate the Molar Mass:
- Molar mass of N₂O₄ = (2 × Atomic mass of N) + (4 × Atomic mass of O)
- Molar mass of N₂O₄ = (2 × 14.01 g/mol) + (4 × 16.00 g/mol)
- Molar mass of N₂O₄ = 28.02 g/mol + 64.00 g/mol
- Molar mass of N₂O₄ = 92.02 g/mol
Which means, the molar mass of N₂O₄ is 92.02 g/mol. This value means that one mole of N₂O₄ weighs 92.02 grams That's the whole idea..
Converting Grams of N₂O₄ to Moles
Now that the molar mass of N₂O₄ is known, you can convert the given mass of 76.3g of N₂O₄ to moles using the following formula:
Number of moles = Mass (g) / Molar mass (g/mol)
-
Plug in the Values:
- Mass of N₂O₄ = 76.3 g
- Molar mass of N₂O₄ = 92.02 g/mol
-
Calculate the Number of Moles:
- Number of moles of N₂O₄ = 76.3 g / 92.02 g/mol
- Number of moles of N₂O₄ ≈ 0.829 mol
Which means, there are approximately 0.That said, 829 moles of N₂O₄ in 76. 3g of N₂O₄.
Step-by-Step Calculation Summary
-
Determine the molar mass of N₂O₄:
- Nitrogen (N): 14.01 g/mol
- Oxygen (O): 16.00 g/mol
- Molar mass of N₂O₄ = (2 × 14.01) + (4 × 16.00) = 92.02 g/mol
-
Use the formula to convert mass to moles:
- Moles = Mass / Molar mass
- Moles of N₂O₄ = 76.3 g / 92.02 g/mol ≈ 0.829 mol
Practical Applications and Significance
Understanding how to convert between mass and moles is essential for various applications in chemistry, including:
- Stoichiometry: Moles are fundamental to stoichiometric calculations, which are used to determine the amounts of reactants and products in chemical reactions.
- Solution Preparation: When preparing solutions of specific concentrations, you need to know the number of moles of solute required.
- Gas Laws: The number of moles of a gas is used in the ideal gas law (PV = nRT) to relate pressure, volume, temperature, and the amount of gas.
- Chemical Analysis: In quantitative analysis, moles are used to determine the composition of substances.
Common Mistakes to Avoid
When converting between mass and moles, make sure to avoid common mistakes:
- Incorrect Molar Mass: Double-check the molar mass calculation. make sure you are using the correct atomic masses from the periodic table and that you have accounted for the correct number of atoms of each element in the chemical formula.
- Unit Confusion: see to it that the units are consistent. Mass should be in grams (g), and molar mass should be in grams per mole (g/mol).
- Calculation Errors: Be careful with your calculations, especially when dividing or multiplying. It’s always a good idea to double-check your work.
- Forgetting Significant Figures: Pay attention to significant figures in your calculations. The final answer should be reported with the appropriate number of significant figures based on the given data.
Examples of Related Calculations
Here are a few examples of related calculations to further illustrate the concept:
Example 1: Calculating Moles of Water in 100g of H₂O
-
Molar mass of H₂O:
- Hydrogen (H): 1.01 g/mol
- Oxygen (O): 16.00 g/mol
- Molar mass of H₂O = (2 × 1.01) + 16.00 = 18.02 g/mol
-
Convert mass to moles:
- Moles of H₂O = 100 g / 18.02 g/mol ≈ 5.55 mol
Example 2: Calculating Mass of 0.5 Moles of Carbon Dioxide (CO₂)
-
Molar mass of CO₂:
- Carbon (C): 12.01 g/mol
- Oxygen (O): 16.00 g/mol
- Molar mass of CO₂ = 12.01 + (2 × 16.00) = 44.01 g/mol
-
Convert moles to mass:
- Mass of CO₂ = 0.5 mol × 44.01 g/mol = 22.005 g ≈ 22.0 g
Example 3: Calculating Moles of Sodium Chloride (NaCl) in 58.44g of NaCl
-
Molar mass of NaCl:
- Sodium (Na): 22.99 g/mol
- Chlorine (Cl): 35.45 g/mol
- Molar mass of NaCl = 22.99 + 35.45 = 58.44 g/mol
-
Convert mass to moles:
- Moles of NaCl = 58.44 g / 58.44 g/mol = 1 mol
Advanced Concepts: Stoichiometry and Chemical Reactions
Once you are comfortable converting between mass and moles, you can apply these concepts to more complex stoichiometric problems. Stoichiometry involves using the relationships between reactants and products in a chemical reaction to determine the amounts of substances consumed or produced It's one of those things that adds up..
Balancing Chemical Equations
The first step in any stoichiometric calculation is to confirm that the chemical equation is properly balanced. A balanced equation shows the correct mole ratios of reactants and products.
Here's one way to look at it: consider the reaction between hydrogen gas (H₂) and oxygen gas (O₂) to produce water (H₂O):
2H₂ + O₂ → 2H₂O
This balanced equation indicates that 2 moles of H₂ react with 1 mole of O₂ to produce 2 moles of H₂O.
Mole Ratios
Mole ratios are derived from the coefficients in the balanced chemical equation. They are used to convert between the moles of different substances in the reaction.
In the above example, the mole ratio between H₂ and H₂O is 2:2 (or 1:1). What this tells us is for every 2 moles of H₂ that react, 2 moles of H₂O are produced. Similarly, the mole ratio between O₂ and H₂O is 1:2, meaning that for every 1 mole of O₂ that reacts, 2 moles of H₂O are produced That's the part that actually makes a difference. But it adds up..
Limiting Reactant
In many chemical reactions, one reactant is completely consumed before the other reactants. This reactant is called the limiting reactant because it limits the amount of product that can be formed. To determine the limiting reactant, you must compare the mole ratios of the reactants to the stoichiometric ratios in the balanced equation.
Percent Yield
The theoretical yield of a reaction is the amount of product that can be formed based on the stoichiometry of the reaction, assuming that the reaction goes to completion and there are no losses. That said, in practice, the actual yield of a reaction is often less than the theoretical yield due to various factors, such as incomplete reactions, side reactions, and losses during purification.
The percent yield is a measure of the efficiency of a reaction and is calculated as follows:
Percent Yield = (Actual Yield / Theoretical Yield) × 100%
The Importance of Accurate Molar Mass Values
The accuracy of molar mass values is crucial for precise chemical calculations. Molar masses are typically determined using the atomic masses provided on the periodic table. These atomic masses are based on the weighted average of the masses of the naturally occurring isotopes of each element And that's really what it comes down to. Surprisingly effective..
Isotopes and Atomic Mass
Isotopes are atoms of the same element that have different numbers of neutrons. Because isotopes have different masses, the atomic mass of an element is the weighted average of the masses of its isotopes, taking into account their natural abundance.
Take this: carbon has two stable isotopes: carbon-12 (¹²C) and carbon-13 (¹³C). Carbon-12 is much more abundant than carbon-13, so the atomic mass of carbon is very close to 12.01 amu.
The Role of Mass Spectrometry
Mass spectrometry is a powerful analytical technique used to determine the masses of atoms and molecules with high precision. In mass spectrometry, a sample is ionized, and the ions are separated according to their mass-to-charge ratio. The resulting mass spectrum provides information about the isotopic composition of the sample, which can be used to determine the atomic and molecular masses with great accuracy.
Concluding Thoughts
Converting between mass and moles is a fundamental skill in chemistry that is essential for understanding stoichiometry, solution preparation, gas laws, and chemical analysis. By following the steps outlined in this guide and avoiding common mistakes, you can confidently perform these calculations and apply them to a wide range of chemical problems. That said, understanding the concepts of molar mass, mole ratios, limiting reactants, and percent yield will further enhance your ability to analyze and interpret chemical reactions. Accurate molar mass values, determined using techniques like mass spectrometry, are crucial for precise chemical calculations.